From custom polymer design and synthesis to formulation of your hydrophobic API
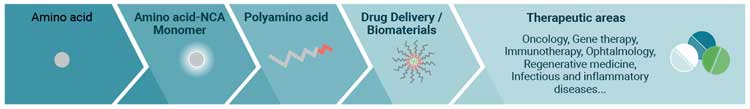
We specialize in creating delivery systems for different active ingredients, like small molecules, oligonucleotides, antibodies, and proteins. Our team includes experts known for their skills in NCA chemistry and Polyaminoacid (PAA) technology.
What sets us apart is our ability to design custom excipients and develop specific formulations to suit your needs. We use polymers as a base for pre-formulation, incorporating ingredients like TPGS, lipids, PEGylated polymers, and polyaminoacid (PAA).
Trust us to tailor-make solutions for your application, ensuring effective and precise delivery of your active payload. Explore the possibilities with Our Offer in Drug Delivery Services!
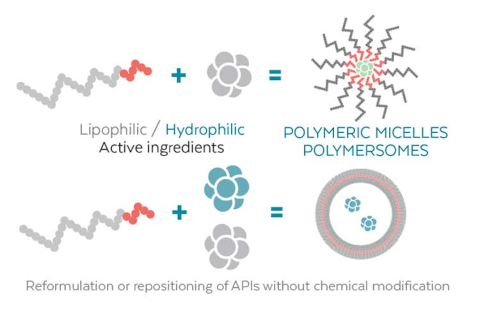
Micelles, Emulsions, PEG-polymer or PAA Nanoparticles, Lipid Nanoparticles (LNP), Polymersomes, Liposomes, Nanogels
You have delivery issues with your API, we have a solution!
The power of PAA technology elevating drug delivery
The encapsulation of APIs into our polymeric nanoparticles offers a multitude of benefits:
- Improved Solubility of poorly water-soluble compounds
- Enhanced Bioavailability
- Reduced Toxicity
- Controlled Release
- Active Targeting of specific tissues or cells
Amino acid based biopolymer diversity for innovatives therapies
A unique integrated solution
- Talented & multidisciplinary R&D team
- Broad network for access cutting-edge technologies
- State-of-the-art / GLP formulation and analytical equipment
- Custom PAA synthesis by Ring Opening Polymerization (ROP) & tailor-made pre-formulation services
- Integrated NCA supply (Phosgene expert, industrial scale)
Unmatched capabilities
- 50 years in large scale manufacturing of drugs substances and excipients under GMP
- Process development and scale up (GMP Kilolab and Pilot)
- Analytical development & QC
- Preclinical, clinical and commercial batches of drug substance and excipient
- 3 FDA & EU qualified facilities (France)
- Full regulatory support (CMC-DMF, Documentation)